|
10/30/2023 0 Comments Dental impression meaning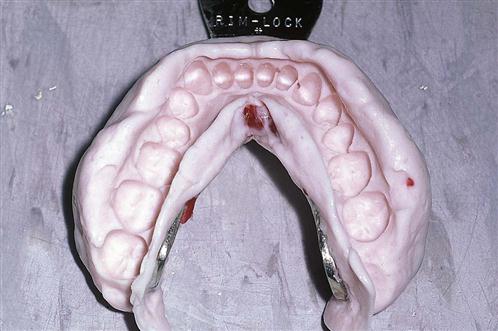
While there are 5 types of impression materials commonly used in dentistry today (e.g., hydrocolloids, polysulfide, addition silicone, polyether, and vinyl-polyether hybrids), only 3 are commonly used for final impressions: polyether, vinyl-polyether hybrids, and polyvinyl siloxane. No impression material meets all of these requirements, but significant improvements have been made. An ideal impression could be poured multiple times, without losing accuracy. Disinfection should not reduce surface detail or accuracy. 
Desirable features also include long working time, short setting time, and a long shelf life. :max_bytes(150000):strip_icc()/GettyImages-944490598-f3b3bc78547f4217a4ef8b9846e91450.jpg)
It must be biocompatible, non-toxic and have an acceptable odor and taste. An ideal impression material should also demonstrate excellent detail reproduction, good tear strength, and no distortion when removed from the mouth. The color of the impression material should be saturated enough to detect whether the prepared tooth margin is captured. It should not shrink during polymerization, shipping or storage and should have excellent flow. An accurate impression is dependent upon proper technique and optimal impression material characteristics.Īn ideal impression material should have many features. Impression materials are used to obtain an accurate replica of hard and soft oral tissues. Alginate is also an example of imbibition because it will absorb water if soaked in it.Elastomeric impression mat erials are used in a variety of dental procedures, and improved delivery systems and materials with better properties have made the impression technique more predictable (Figure 1). The opposite process of syneresis is imbibition, which is the process of a material absorbing water molecules from the surroundings. Gels formed from agarose are prone to syneresis, and the degree of syneresis is inversely proportional to the concentration of the agarose in the gels. For this reason, many dental impression companies strongly recommend to pour the dental cast as soon as possible to prevent distortion of the dimension of the teeth and objects in the impression. Due to this process, the impression shrinks a little and therefore its size is no longer accurate. In dentistry, syneresis is the expulsion of water or other liquid molecules from dental impression materials (for instance, alginate) after an impression has been taken. Additionally, it creates unsightly moisture pockets within baked custard dishes, such as flan or crème brûlée. It also causes emulsified sauces, such as hollandaise, to "break" ("split"). It creates weeping in scrambled eggs, with dry protein curd swimming in the released moisture. This process is responsible for transforming juicy rare steak into dry steak when cooked thoroughly. The hard protein shell pops, expelling the moisture. In cooking, syneresis is the sudden release of moisture contained within protein molecules, usually caused by excessive heat, which over-hardens the protective shell. The splitting of the bond between residues 105 and 106 in the κ-casein molecule is often called the primary phase of the rennet action, while the phase of coagulation and syneresis is referred to as the secondary phase. This process is usually referred to as the phase of coagulation and syneresis. Bonds between hydrophobic sites start to develop and are enforced by calcium bonds, which form as the water molecules in the micelles start to leave the structure. In the processing of dairy milk, for example during cheese making, syneresis is the formation of the curd due to the sudden removal of the hydrophilic macropeptides, which causes an imbalance in intermolecular forces. Syneresis has also been proposed as the mechanism of formation for the amorphous silica composing the frustule of diatoms. A household example of this is the counterintuitive expulsion of water from dry gelatin when the temperature increases. Syneresis can also be observed when the amount of diluent in a swollen polymer exceeds the solubility limit as the temperature changes. Another example of syneresis is the collection of whey on the surface of yogurt. Syneresis (also spelled 'synæresis' or 'synaeresis'), in chemistry, is the extraction or expulsion of a liquid from a gel, such as when serum drains from a contracting clot of blood. 
JSTOR ( February 2015) ( Learn how and when to remove this template message).Unsourced material may be challenged and removed.įind sources: "Syneresis" chemistry – news Please help improve this article by adding citations to reliable sources. This article needs additional citations for verification.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
